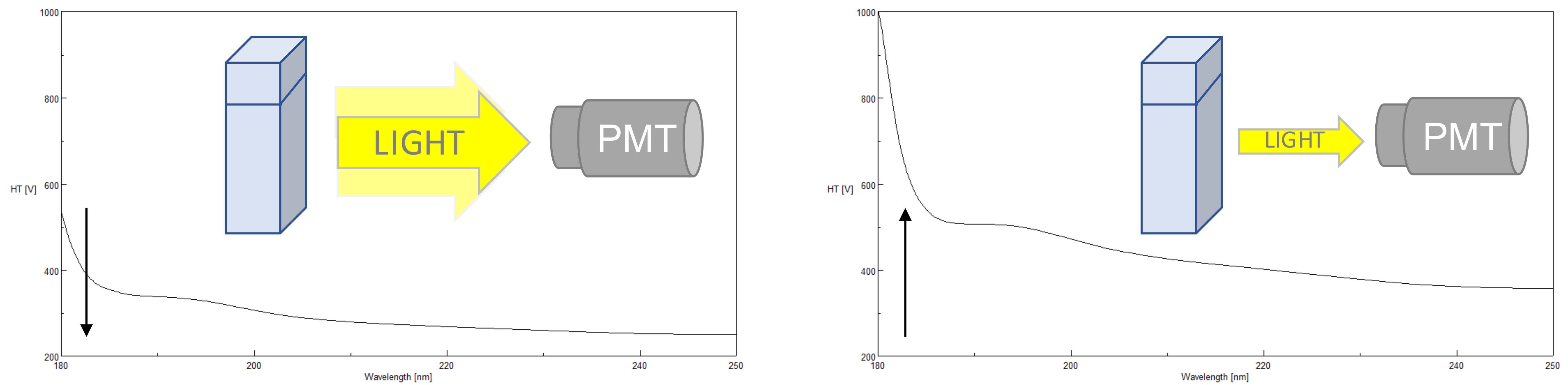
So, if the aim of the experiment is to measure a spectrum in the far UV, for example, to measure a protein spectrum between 180nm and 250nm to determine its secondary structure, then yes nitrogen flushing is necessary as we need to go below 195 nm. However, above 195 nm the high voltage is similar, so it means the oxygen is not absorbing light and nitrogen purging, therefore, becomes useless for the measurement. Thanks to nitrogen, the CD signal can be recorded accurately below 195 nm. Spectra are shown in figure 2.įigure 2: High Voltage scan: blue (without N 2), and red (with N2)īelow 195 nm we can clearly see the importance of nitrogen flushing. To see the real effect of oxygen one can thus run a lamp spectrum with nitrogen flushing (so without oxygen in the light path) or without nitrogen (so with oxygen present in the light path). And usually, data obtained with a high voltage above 550-600V are considered doubtful. So, the less light you have, the higher the high voltage is. The high voltages used by the CD detector during measurements are directly linked to light levels. This is because oxygen absorbs light in the far UV and the deeper you go in the UV the more it absorbs (see figure1).
But it is also true that nitrogen use should be limited to far-UV needs and not used with near UV, visible, or NIR regions. The main reason given is the need to remove oxygen and to reduce its light absorption in the far-UV range. Why waste money on nitrogen for Circular Dichroism?įor decades, some Circular Dichroism (CD) instrument manufacturers have made nitrogen (N 2) flushing of optics an absolute requirement for CD measurements, but is it always really necessary? The answer is no.

